Brain Injury Reversed
“Kentuckiana MD Brings New Hope for the reduction and or reversal of symptoms in patients with Parkinson, ALS, MS, Stroke, Brain Injury, and Depression”
Disclaimer:
Results variable.
Results NOT guaranteed.
Symptom improvement is NOT guaranteed and guarantees are not made or implied.
Non-FDA approved therapies are not designed to treat, cure or diagnose any particular illness, but rather to support self-healing mechanisms.
Consultation: $400
Evaluation and blood tests are needed before treatment can be approved.
INSURANCE NOT ACCEPTED since these management strategies are considered experimental.
If accepted for treatment, cost range is $9,000 – $30,000 depending on management strategies recommended and selected.
Innovative Treatment for reversing symptoms of Parkinson, CRPS, ALS, Stroke & Traumatic Brain Injury
Our treatment approaches transform lives from dysfunctional to functional. To see what is possible review videos shown below to see the amazing recoveries that may occur after treatment even in patients with little function 10-15years after their stroke or brain injury. DISCLAIMER: Results vary from person to person. Videos should not be taken as a guarantee of your results. Some patients receive NO BENEFIT with treatment.
Mr. Ed Wilks visits us before stroke treatment. This is the first part of his evaluation.
Mr. Ed Wilks had a stroke. This is the second part of his evaluation before treatment.
Most patients and physicians believe that after a stroke or traumatic brain injury, patients will NOT recover any new brain or neurological function after 2 years from the date of their original injury. While it is true most patients DO NOT develop any new brain or neurological function after two years, there are innovative therapies for Stroke, Traumatic Brain Injury [TBI], Alzheimer’s Dementia and Intractable Spine Pain that are restoring function and have transformed the treatment of these patient’s injuries.
Our treatment approaches are changing lives from hopeless to hopeful! Now patient’s who are even 10-20 years past their injury are recovering a tremendous amount of brain, neurological and or nervous system function.
Discover what is possible at
Kentuckiana Integrative Medicine:
Louis Meier, 75-year-old Retired Mechanic who came to our office in a motorized wheelchair with advanced Parkinson’s, Lumbar Fusion, Severe Back Pain, Severe Hip Pain, Shuffling gate, Recurrent Falling for 6 months.
60 Minutes Australia documents a New England Teacher with Stroke, abnormal walking and expressive aphasia [inability to speak] who recovers fluid speech, reading and normal walking.
There are additional testimonials at www.regenmedky.com
We are fortunate to have treatments available in Kentuckiana [Louisville KY–Jeffersonville IN] thanks to the training received by Dr. Rafael F. Cruz MD from Kentuckiana Integrative Medicine – Kentuckiana Neuro Recovery Center.
Dr. Cruz’s training is in Emergency Medicine, Anesthesia – Pain Management and Integrative-Functional Medicine. He believes in blending the best of both traditional conventional medicine with the best of natural medicine integrating the best therapies from both sides of medicine. These management strategies are available at almost no other medical centers in any neighboring states.
After Dr. Rafael Cruz’s father, Dr. Rafael Cruz Sr. suffered a cervical cord spinal stroke due to surgical complications, his father did the standard physical therapy but made modest to little progress. Dr. Cruz Sr. was left with weak hands, arms & legs, and poor balance. Several years later, desperate to find a solution to help his 82 year old father, a formerly active surgeon and tennis player [who retired and played singles tennis at 79 years old], Dr. Cruz learned and trained on the use of several innovative approaches for the treatment of neurological disorders.
After extensive additional training, Dr. Cruz Jr. was ready to treat his father. Dr. Cruz’s sister, a family doctor in New Jersey, came to Jeffersonville to assist her brother. Before treatment, Dr. Cruz Sr. could not get up from a chair without assistance. He could not climb a staircase without a great deal of assistance and support from both of his children. He could not remember the five words from the pretreatment memory test.
The changes after treatment were truly remarkable. Within 20 minutes, Dr. Cruz Sr.’s memory was improved as he remembered a few of the five words from the earlier memory word test. Three hours later, he could lift himself out of an arm chair without assistance and he could climb a stair case of 10 steps – very, very slowly, but without any help. At dinner that evening, Dr. Cruz Sr. remembered all of the five words from the pretreatment memory test. There was no question in either sibling’s mind that their father was markedly improved – the changes were undeniable, pronounced, fast and dramatic.
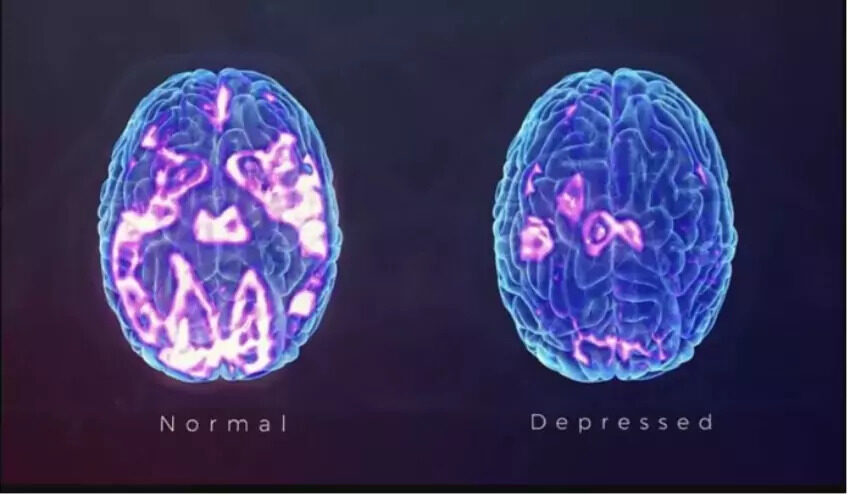
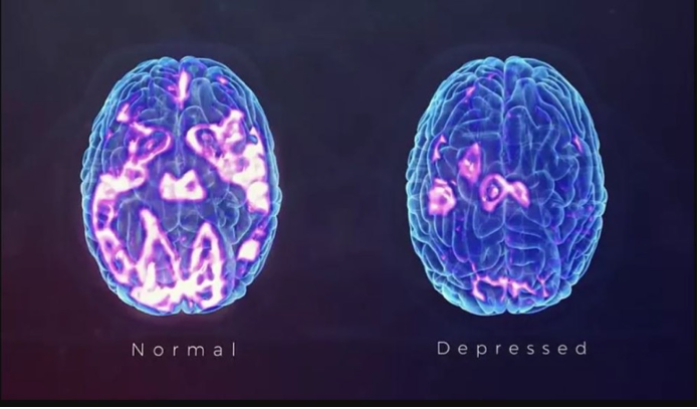
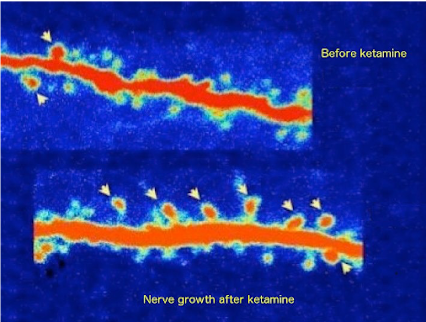
Dr. Cruz is able to help patients with a variety of FDA approved and NON-FDA approved therapies such as: IV Sodium EDTA chelation, IV Plaquex, IV Glutathione, IV Ketamine, IV Magnesium, IV Vitamin Mega C Meyers cocktail which are also helpful with Chronic Fatigue, Fibromyalgia, Neuropathic Pain, CRPS, OCD, PTSD, Anxiety, Suicidal Thinking, Migraines and Depression. Many treatments improve brain function. Etanercept, Ketamine, B vitamins and Magnesium are helpful to brain function. The images below depict functional brain improvement using Ketamine.
Ketamine stimulates growth of nerves and nerve connections creating enhanced neural pathways.
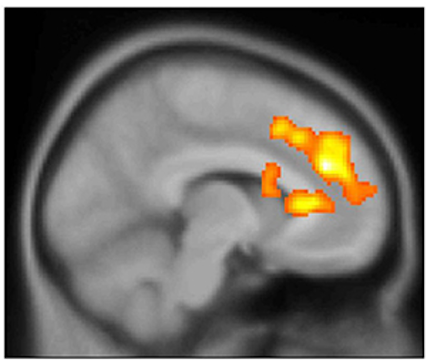
PET scans revealed that ketamine rapidly restored bipolar depressed patients’ ability to anticipate pleasurable experiences by boosting activity in the dorsal anterior cingulate cortex (yellow) and related circuitry. Picture shows PET scan data superimposed on anatomical MRI.38
Source: Carlos Zarate, M.D., Experimental Therapeutics and Pathophysiology Branch, NIMH
In spite of early controversy, Peri-Spinal Etanercept was approved by a federal judge in Florida and one in California as within the standard of care based on the large amount of research results and data.

These treatments are offered by very few physicians across the country. Dr. Rafael Cruz was fortunate enough to train with the inventor of these patented treatments. Dr. Cruz offers other treatments that are may be helpful to patients with Parkinson’s, TBI and Stroke. Call at Kentuckiana Integrative Medicine 812-913-4416 or 812-282-1575 for a consultation.
*Disclaimer: Individual results vary, not all patients respond. Additional treatments may be necessary to maintain the clinical response. Treatment for these indications is innovative (“off-label” and not FDA approved).
“Ketamine, given intravenously, might be the most important breakthrough in antidepressant treatment in decades.” Thomas Insel Director, National Institute of Mental Health
“I had come to believe that my depression was a terminal illness. But the so-called party drug [Ketamine] may have saved my life.” Alice Levitt-Medical Writer and Editor
Science that Feels Miraculous
“It’s magic,” said Dr. Rene Alfaro, an ophthalmologist from Mexico, of the single-dose injection that almost instantly restored much of his wife’s movement and cognition more than a year after an aneurysm and subsequent stroke incapacitated her left side. “It’s like a click.” Within minutes of receiving the injection on Monday, Anna Alfaro walked without a cane, got up from a chair more easily and regained sensation in her left arm. Hours later, when some of her left eye vision returned, she could see the phone clearly enough to dial a number for the first time since her stroke.
Her response is not unique.
Louisville resident and successful grocery owner, John Campisano suffered a stroke during his heart bypass surgery. He awoke with expressive aphasia [unable to speak or pronounce words] and very poor balance. Expressive aphasia is a condition in which the patient understands words and pictures, but is unable to pronounce words to express his or her needs or what he or she sees in words and numbers.
Mr. Campisano consistently responded to his yearly treatments with “my mind is clear,” improved speech fluidity, improved word and picture recognition and improved balance. After one of his treatments just before a family Christmas party, Mr. Campisano shocked and delighted his daughter Karen, his brothers and cousins entertaining them with stories from their past.
[Note: Mr. Campisano presented to Dr. Cruz 20 TWENTY years after his STROKE!!! This means even though conventional medicine believes no improvement is possible 2 years after Stroke or TBI injury; there is hope even 20 YEARS LATER!!!] His daughter Karen Campisano is our practice manager.
The PSE peri-spinal etanercept treatment consists of a single injection given in the soft tissues of the cervical spine, followed by a short wait of several minutes to allow the medication to get to the brain. That’s all it took for hundreds of stroke and traumatic brain injury patients from South Florida and nationwide to reverse years of debilitation. Now they are walking more steadily, reading more easily, concentrating better, speaking more clearly and regaining use of once-rigid limbs – long after giving up hope that their bodies would ever respond.
The medicine in the 25-milligram injection that provides renewed independence is etanercept [a rheumatoid arthritis medication sold under the brand name Enbrel among others]. This treatment approach represents a “radical breakthrough” in the management of chronic neurological dysfunction.
The medicine in the 25-milligram injection that provides renewed independence is etanercept [a rheumatoid arthritis medication sold under the brand name Enbrel among others]. This above treatment approaches represent “radical breakthroughs” in the management of chronic neurological dysfunction.
According to the above study, of the total patients studied, more than 80 percent saw improvements in their ability to walk; more than 80 percent had less spasticity; and more than 85 percent exhibited improved motor function.
This is an impressive treatment response rate because it means that out 629 patients; 503 patients saw improvements!
Improvements also were recorded in many patients’ range of motion, pain and cognition, as well as their ability to *speak, *see, *swallow, *concentrate and *maintain bowel & bladder control. The benefits reportedly are long-lasting in many patients. To promote the potential for long lasting results, patients must engage in physical therapy at home and or physical activity so that their newly discovered neuro-plastic changes can have an opportunity to wire and fire together in a more permanent manner. The old adage of “use it or lose it”, definitely holds true. According to the first patients treated in November 2010, many continue to enjoy improved mobility and cognition.
Note with all of the above success, there are an approximate 20% of patients who receive NO benefit at all from this therapy so patients and family must accept the very real possibility that they may not improve.
Hollywood, Fla., resident Jennifer Cody said the treatment gave her 59-year-old father, a former outdoor maintenance man, renewed independence 18 years after his stroke.
“Within five minutes, her father was saying how clear his head was,” Cody said. “It had always been so cloudy.”
Now her father talks without slowed speech, he’s confident enough to drive on the highway, he can roll down the window in his car, and he no longer drags his left leg behind him.
“He does one-legged squats now,” [18 YEARS AFTER A STROKE] Cody said, with a laugh. “For them to be able to do something like that for someone who had a stroke (so many) years ago, it’s incredible. I’m telling everyone I know.”
Though most people are satisfied with their improvement after a single dose treatment, a second treatment dose after one month can be administered if patients saw some improvement but wanted an opportunity for even more improvement. About 25% patients, including Cody’s father, choose to receive a second injection.
“These results represent a sea change in the therapeutic possibilities for stroke and TBI patients,” Steven Ralph, associate professor at Griffith University School of Medical Science in Australia, said in an article produced by Adis’ parent company, Springer Science + Business Media. “Rarely do we see such a radical breakthrough in medical treatment as this for stroke.”
Numerous scientific and medical journals around the world – have found that this therapeutic technique is also beneficial for treating sciatica and Alzheimer’s disease.
Etanercept – better known by its brand name Enbrel – has proven effective in neutralizing TNF – Tumor Necrosis Factor, the chemical that causes inflammation in the body, and has Food and Drug Administration approval for treating rheumatoid arthritis and psoriasis.
Enbrel’s makers warn of side effects from continued use, including headaches, upper respiratory infection and, in severe cases, immune system impairment, risk of cancer, heart failure and blood and nervous system problems. Stroke and TBI patients undergoing this treatment are receiving only one, maybe two doses of etanercept so it is rare for patients to experience side effects.
Because the therapy’s “off-label” use of etanercept is not FDA-approved, this treatment is not covered by Medicare or other insurance plans.
For Lantana, Fla., resident Martha Levine, 88, it meant being able to feed herself again. More than THREE years after a stroke left the retired clinical social worker with a severely weakened left side.
Martha’s gait-ability to walk- immediately grew steadier and quicker, and she was able to think more quickly and clearly, said her husband, Irvin Levine.
Still dependent on a walker and, sometimes, a wheelchair, her improvements were important but not dramatic, her husband said. “When you stop to think she had nothing before and she’s a little better now, to us, it was a big difference,” he said. “Even though it’s slight, we’re thankful for it.”
Anna Alfaro’s quick, dramatic improvements had her husband in tears. The couple, with their 13-year-old son in tow, flew in seeking a glimmer of hope after doctors in Mexico City had assured them nothing more could be done for her.
“The real important thing is the daily things, the simple things,” said Anna Alfaro, who had been dependent on a cane and no longer able to manage her husband’s glaucoma diagnostic center. “For me, this is the difference in my life. I’m going back to work.”
Who Is A Candidate For Treatment?
The vast majority of stroke and traumatic brain injury survivors living at home or in a rehab center are candidates for our therapies. The best patients are those with some residual function in their mobility and cognition.
Patients who would NOT qualify for treatments are PATIENTS DIAGNOSED WITH:
- Active cancer
- Bleeding Disorder
- Blood Disorder-
- Lymphoma
- Active Current Congestive Heart Failure Symptoms
- Active Tuberculosis (Those with a history of +PPD test or history of Tuberculosis may still be candidates if properly treated and have a normal Quantiferon Gold Test)
- Severe immune suppression with organ transplant medications
- Uncontrolled diabetes
- HIV
- Syphilis
- Hepatitis
- Hospitalized patients NOT yet discharged
- Comatose patients
Consultation: $400.
INSURANCE NOT ACCEPTED since these management strategies are considered experimental. If accepted for treatment cost is $9,000 – $30,000 depending on management strategies recommended and selected.
Those interested will want to submit records to kimregenmed@gmail.com with most recent notes from treating family doctor, treating neurologist, or treating neurosurgeon, written reports of all labs, MRI scans, CT scans, List of diagnoses, current list of medication, supplements, and allergies. Patients will require new blood work to screen for immune suppression, uncontrolled diabetes, anemia, abnormal liver function, abnormal kidney function, Hepatitis, HIV, RPR consisting of CBC, CMP, HA1C, Quantiferon Gold, etc. If you email information, you should know it could be discovered by others since the internet is not 100% secure. If you email our office, you must accept the risk of your information possibly and inadvertently being disclosed to others and you must agree to not hold this office or its representatives responsible which occurs as soon as you email us. If you decide to email our office, you should call the office when you send your information.
Testing can be conveniently obtained from LabCorp or Quest. Testing is not always covered by insurance
For more information, contact Kentuckiana Integrative Medicine-Kentuckiana Neuro Recovery Center at 812-913-4416 located next to Louisville, KY at 405 East Court Av. Jeffersonville, IN 47130, or go to www.regenmedky.com
Karen Campisano Manager: Kimregenmedky@gmail.com
Innovative Treatment for Stroke & Traumatic Brain Injury
Some patients experienced amazing recoveries that occur soon after treatments even in patients who were injured 10 years earlier.
Most patients and physicians believe that 2 years after a stroke or traumatic brain injury, most patients will NOT recover any new brain or neurological function after 2 years from the date of their original injury. While it is true most patients DO NOT develop any new brain or neurological function after two years, there are innovative therapies for Stroke, Traumatic Brain Injury [TBI], Alzheimers Dementia and Intractable Spine Pain that are restoring function and have transformed the treatment of these patient’s injuries.
Brain Injury Treatments are changing lives from hopeless to hopeful! Now some patients who are even 10-20 years past their injury are recovering a tremendous amount of brain, neurological and or nervous system function.
Results vary from spectacular or miraculous to NO response or benefit. When patients respond to these treatments, they respond within minutes and or over 24-48 hours. Function, memory, vision, strength, mood and balance frequently improve very quickly.
These treatments are offered by very few physicians across the country. Dr. Rafael Cruz MD was fortunate enough to train with some of the most innovative clinics & physicians in the country. Dr. Cruz is now offering MANY of these therapies at Kentuckiana Integrative Medicine. Call 812-913-4416 or 812-282-1575 for a consultation.
Disclaimer: Individual results vary, not all patients experience benefit from these therapies. Additional treatments may be necessary to maintain the clinical response. Treatment for these indications is innovative (“off-label” and not FDA approved)
Here’s What To Do Next
- Call 812-913-4416 or 812-282-1575 to schedule a consultation.
- Complete lab work at either Quest or Lab Corps. DOWNLOAD LAB REQUEST
- Complete the patient information/history form. COMPLETE INTAKE FORM HERE



